BCS Trial Outline
The figure below illustrates the temporal sequence of British Cold Study (BCS) study procedures. See the table below for a more detailed description of the measures taken during each phase of the study.
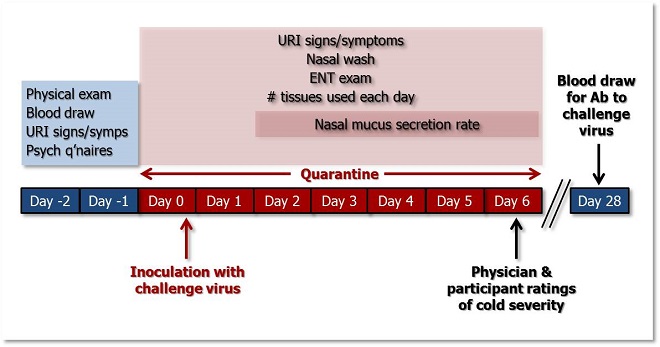
Figure 1. Temporal sequence of BCS study activities. >>Download PowerPoint Slide<<
BCS Measures by Study Phase
| Study Phase | Measures Collected |
|
1-2 days Pre-Viral Challenge (Days -1 & -2)* |
• Physical exam • Blood draw: o Complete Blood Count (CBC) with differential and serum chemistry; o total & viral-specific serum Ab; o cotinine • Clinician reports: Upper Respiratory Illness (URI) signs and symptoms • Number of tissues used each day • Nasal wash: o total Ab (protein, IgA, & IgE); o specific Ab (IgA) • Nasal mucus secretion rate |
|
End of Quarantine Day 0 |
Inoculation with Virus (i.e., Viral Challenge) |
|
Quarantine Days 0 through 6 |
• Clinician reports: URI signs and symptoms • Participant reports: URI symptoms • Nasal wash: specific IgA • Ears, nose, and throat (ENT) exam • Number of tissues used each day • Nasal mucus secretion rate (Quarantine Days 2-6) |
| Quarantine Day 6 |
• Physician and participant ratings of cold severity |
|
4 weeks Post-Challenge |
• Blood draw: total and viral-specific serum Ab; cotinine |
*Participants in the British Cold Study (in contrast to the three Pittsburgh Cold Studies and the Pittsburgh Mind-Body Center Cold Study) were quarantined in the Common Cold Unit for two days prior to inoculation with the challenge virus.
Study Questionnaire Sets
Questionnaire Set 1
Life Events List
Interpersonal Support Evaluation List
Mood States Scale, Part A
Questionnaire Set 2
Perceived Stress Scale
Fleming & Watts Self Esteem Scale
Mood States Scale, Part B
Social Network Index
Recent Cold History
Questionnaire Set 3
Spheres of Control Scale
Demographics
Physical Activity Questionnaire
Alcohol Consumption Questionnaire
Eating Habits Questionnaire
History and Current Status of Smoking
Sleep