Gel Beads and Worms
One type of polymer commonly used in the food industry is called Sodium Alginate. Sodium alginate is a very large polymer, with molecular weights in the range of 1 to 10 million (the molecular weight of water is 18). It is a naturally occurring polymer obtained from the giant kelp (brown seaweed). Kelp is a plant that grows underwater (to as much as 100 feet!) in the oceans along the coastlines of North and South America, New Zealand, Australia and Africa. It is an amazing plant that can grow at the rate of one inch per hour! The kelp plants are cut four times per year and mixed with water and salts to take out or extract the sodium alginate polymer. Sodium alginate is commonly used to thicken and stabilize food mixtures. It is used in foods such as syrups, sauces, cheese products and whipped foods. It is also used in cosmetics and medicines, such as tablets, creams, ointments, shampoo and lotions.
This experiment uses sodium alginate powder to illustrate the concept of cross-linking of polymer chains. Kelp Beads© (and worms) are made from Kelp extract obtained from the Sunnyside Sea Farms in Santa Barbara, California. The dried kelp extract is made up of long kelp sugar chains (called alginates) that have undergone a complex process to remove their Calcium. In nature, Calcium ions react with the sugar chains to bind/crosslink them together. In this experiment, we will combine the Calcium-free alginate solution with a solution of Calcium Chloride to produce gelled beads and strings (called worms!). Alginate gels are quite porous, with many spaces between the bound chains. The resulting gel contains about 1% alginate and 99% water.
SAFETY NOTE: The gel beads and worms are NOT edible. Please DO NOT EAT them!
Supplies needed:
- Colored solutions of Sodium Alginate (Kelp extract) (instructions for preparation included below)
- Aqueous Calcium Chloride solution (instructions for preparation included below)
- 2 Small (3 oz) cups
- Wooden stick or dropper
- Paper plate

Procedure: (reference: Materials from Sunnyside Sea Farms, Santa Barbara, CA)
1. Fill one cup 1/3 full with the calcium chloride solution.

2. Pour enough of the colored sodium alginate solution in the second cup to cover the bottom of the cup.
3. Take a small amount of the sodium alginate solution on the end of a wooden stick or dropper. Let it drip slowly from the stick or dropper into the cup containing the calcium chloride solution. Wait for 20-30 seconds. Remove the beads carefully. Observe and touch the product. Describe its properties. Notice that the beads still contain liquid alginate solution inside the gelled skin.



4. Repeat the step above using more sodium alginate solution. This time pour it in a thin, continuous stream into the calcium chloride solution. Wait 20-30 seconds and pull out the product. Describe its properties.

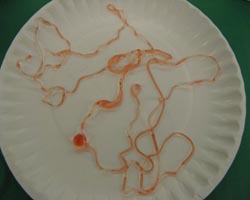
5. Repeat the above step with a sodium alginate solution colored using Tempera® paint. Do you notice any differences between the two colored alginate solutions during the experiment?

6. The beads and worms will become completely gelled if they are left in the calcium chloride solution long enough. The calcium-rich water seeps (diffuses) through the bound chains of the skin, and interacts with the liquid chains inside the bead, continuing to gel the polymers. How much time does it take for your beads and worms to become completely gelled? (A bead 2mm in diameter will take ~ 5 minutes to gel completely, while a 20 mm bead will gel in 10-15 minutes).

Using both red food coloring and blue paint in the alginate experiment illustrates another property of network polymers: porosity. The red food coloring diffuses out of the gel worm and turns the CaCl2 solution red. The blue paint does not! The paint molecules are too large to diffuse out of the pores, so they become trapped in the gel, and the CaCl2 solution stays clear.


Sodium alginate is a very versatile polymer with numerous uses in food manufacture. An example of the use of alginate in food processing is the preparation of onion rings. The onions are chopped, mixed with alginate, starch (another natural polymer!), corn grits, flavors and spices. This mixture is extruded in an onion ring shape and sprayed with a calcium chloride solution. Within 4 seconds, a "skin" forms (due to the calcium/alginate reaction) to hold the onion ring together. The onion ring is then covered with batter, fried and frozen. Advantages to this type of processing include more onion in the extruded onion ring than in the "real" onion rings and the use of small pieces of food that are of good quality, but would otherwise have to be wasted. This process is also used to prepare molded shrimp, chicken, clams, fruit rings and mushrooms.
The alginate gel is also used as a coating for frozen food. By coating the food with this gel, the loss of moisture during the freezing process is minimized, which leads to less freezer burn. The frozen item can remain frozen for a longer time without a decrease in the flavor and quality of the food. Other uses for the alginate gel chemistry include gelled air fresheners, candles, deodorant sticks and dental impressions.
Have your students check the ingredient list of foods at home in their refrigerator, freezer and pantry. Polymer food additives like alginate include gums such as xanthan gum, gellan gum, starches, guar gum, locust bean gum, pectin and gelatin. These materials are all naturally occurring polymers. They are added to food to thicken or gel the product. Only a very small amount of these types of polymers are needed, so they are usually listed at the end of the ingredient list. By examining the ingredient list of foods, how many food items can the student find that contain these polymers?
PREPARATION OF SOLUTIONS FOR THIS EXPERIMENT:
5% solution of calcium chloride, CaCl2
- Dissolve 50 grams of CaCl2 in 1 liter of distilled water.
2% Sodium alginate solution
- Add 6 grams of Keltone® HV (from Sunnyside Sea Farms) to 300 ml of distilled water, while mixing under high shear (a high speed mixer or blender is needed).
- Mix for 15 minutes until all powder disappears.
- Separate a small amount of the solution. Add food coloring to the rest of the solution while mixing.
- Add Tempera paint to the small amount of solution set aside. Choose a different color than the food coloring used above.
Keep the sodium alginate powder and solutions refrigerated at all times, otherwise mold will form. The solution has a shelf life; once it gets thin (lower viscosity) its ability to cross-link is reduced.