Surfaces
Flat Surfaces
Control of surface properties:
Grafting-to:
Grafting-from:
Flat Surfaces
The use of ATRP is particular advantageous in a "grafting from" a flat surface as the thickness of the polymer brush can be precisely controlled by systematic variation of grafting density and DPn of the tethered polymers.(1) (See section on initiation from surfaces) Furthermore, the surface properties can be tuned by the tethering of block copolymers, where the composition and DPn of each polymer segment directly affects the morphology and behavior of the polymer brushes. Modification of surfaces with thin polymer films can be used to tailor the surface properties such as hydrophylicity/phobicity, biocompatibility, adhesion, adsorption, corrosion resistance and friction.
Nanoscale organization of the functional surface can be directed by photolithography and micro and nanoscale printing.(2) The chemical nature of the underlying material becomes hidden by the presence of a film a few Angstroms thick and the interaction of the whole system with the surrounding environment is governed by these coatings.
Control of surface properties:
In addition to "grafting from" a functionalized surface it is also possible to prepare tethered block copolymers by the "grafting to" approach. However, because of kinetic and thermodynamic restrictions, the use of various grafting-onto approaches normally leads to formation of polymer brushes with relatively low grafting density. However this does not mean surface properties are not affected. We have used ATRP to modify surface properties, create nano-patterns and design stimuli-responsive, mainly temperature responsive, materials. Ellipsometry, contact angle, XPS and AFM are used to assess whether tethered (co)polymers possess precise molar mass and composition.
Grafting-to:
The grafting to approach is exemplified by the preparation of a custom designed ABC block copolymer where one block contained a silylating agent. A poly(dimethylsiloxane) (pDMS) macroinitiator was prepared by living anionic ring-opening polymerization of D3 followed by hydrosilation reactions to incorporate a 2-bromoisobutyrate end group suitable for initiation of ATRP onto each active chain end. The ATRP of styrene using the pDMS macroinitiator yielded a diblock copolymer. Chain extension of the pDMS-b-pS macroinitiator with 3-(dimethoxymethylsilyl)-propyl acrylate (DMSA) by ATRP yielded an ABC triblock copolymer. The reactive DMSA segment was covalently attached to silanol groups on a silicon wafer.(3) Due to the low grafting densities inherent to this approach, the tethered copolymer chains retained adequate degrees of freedom enabling segment selective behavior when the surfaces were exposed to various solvents. The resulting tethered ultrathin film can present either a hard surface or a soft-lubricating surface to the environment depending on treatment with segment selective solvents.
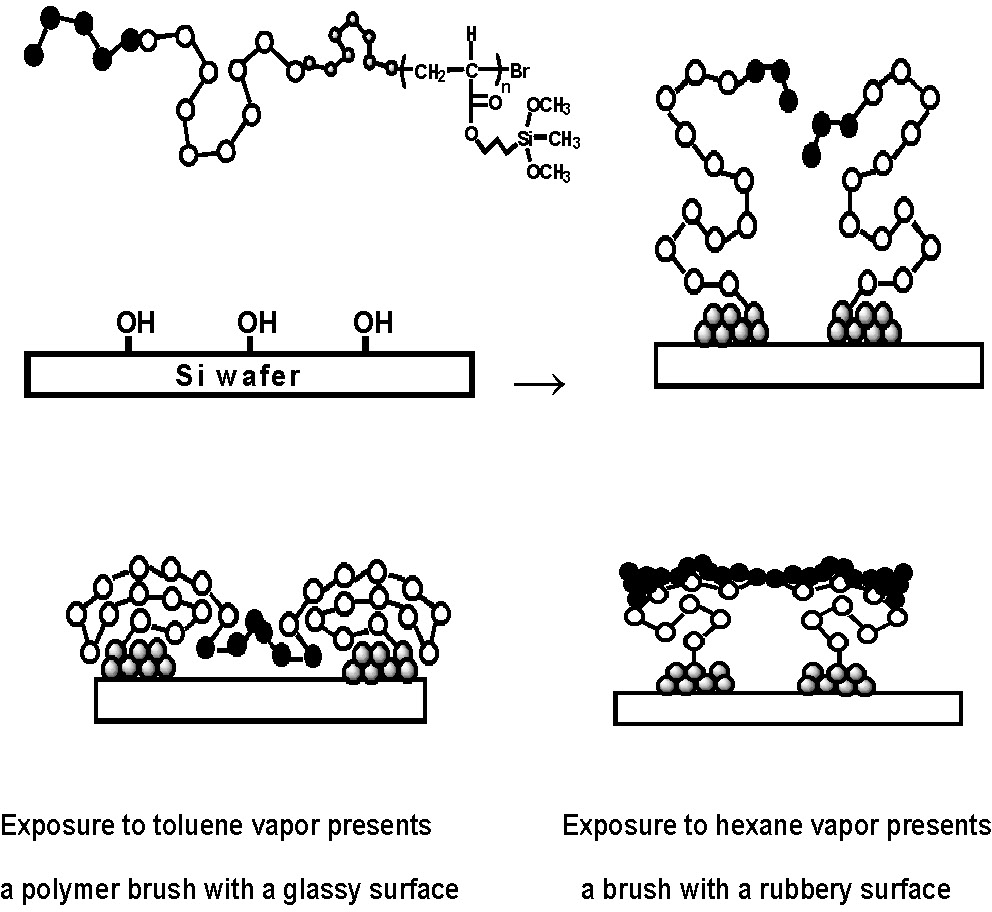
The presentation of either glassy pSt or flexible pDMS segments of the brushes attached to the surface was reversibly controlled by treatment with solvents selected for preferential solvation of each segment.
A similar approach was applied to the synthesis of a diblock copolymer containing poly(N,N-dimethylaminoethyl methacrylate) which was extended with a poly(trimethoxysilylpropyl methacrylate) block. Since pDMAEMA exhibits a lower critical solution temperature (LCST), surface properties of the copolymer ultrathin films could be reversibly controlled by treatment with selective solvents, or temperature. Due to the temperature dependent solubility the tethered chains could change their conformation from extended to globule as the temperature was increased through the LCST.
Grafting-from:
The grafting-from approach, or polymerization from a surface immobilized initiator, generally leads to the formation of a higher graft density of polymer brushes than the grafting-onto approach. By tuning/preselecting reaction parameters; including grafting density, chemical composition or type of substrate, products with different surface properties can be achieved. Silicon wafers are among the most commonly used flat surfaces for these grafting-from reactions and initiators can be attached to both kinds of silicon substrates, namely oxidized (Si-OH) or hydrogen-terminated silicon (Si-H).(4,5) Tethering a chlorosilane (mono- or trichloro) functionalized initiator to an oxidized substrate is the most frequently documented route for the preparation of a surface for a grafting from ATRP.
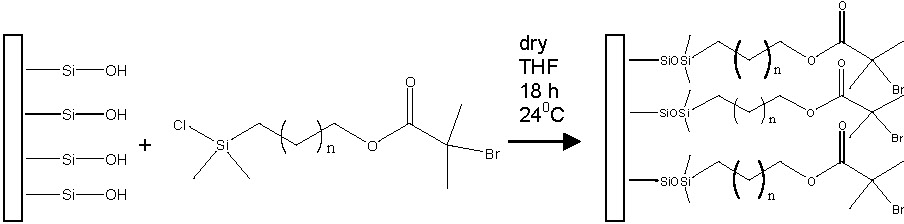
The preparation of block copolymer brushes using a "grafting-from" ATRP approach was first reported by tethering polystyrene-block-poly(t-butyl acrylate) (pSt-b-ptBA) to Si wafers.(5) Hydrolysis of the t-butyl groups yielded a polystyrene-block-poly(acrylic acid) brush demonstrating a versatile approach to tune film properties and hydrophylicity. The concentration of initiating groups on the surface can be varied by varying the molar ratio between two chlorosilanes, one containing an active initiator functionality and the other a "dummy" initiator, each attached through silanol groups to the surface of silicon wafers. For oxidized wafer surfaces, chlorotrimethylsilane was used as a "dummy" initiator and 1-(chlorodimethylsilyl)propyl 2-bromoisobutyrate as an active ATRP initiator.
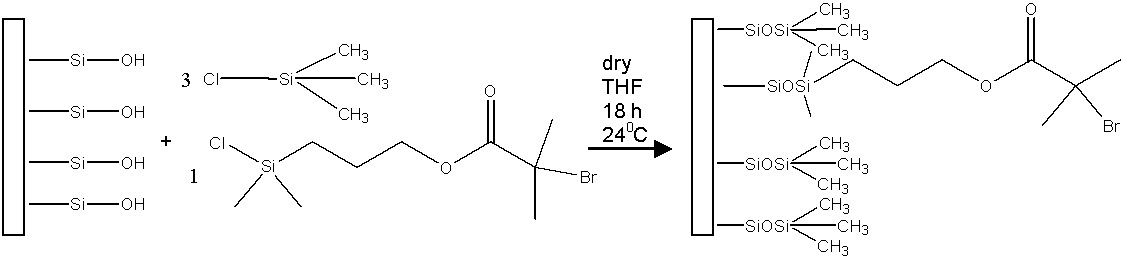
A similar approach was used to attach initiators to/passivate Si-H substrates. A non-functionalized 1-alkene was used as the "dummy initiator" to allow variation of the concentration of initiating groups on the hydrogen-passivated silicon substrate. The initiating groups were attached to freshly hydrogen-passivated silicon wafers by a hydrosilylation reaction which was conducted by exposing the substrate to UV light in the presence of 1- alkene and allyloxytrimethylsilane (TMS). In a second step, the TMS group was converted to 2-bromoisobutyrate ester (an ATRP initiator) using the corresponding acid bromide in the presence of tetrabutylammonium fluoride in dry tetrahydrofuran.
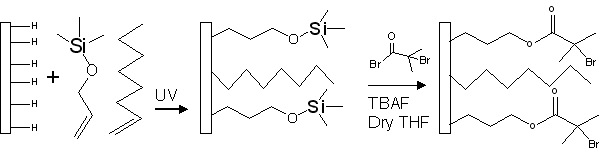
Silicon surfaces modified with ATRP initiators can be transformed to allow for controlled polymerization by RAFT or NMP by use of appropriate ATRC reactions.
When conducting an ATRP from a surface it is recommended that one adds sacrificial initiator to the contacting solution at the beginning of the reaction since this allows controlled chains growth from the start of the reaction. It is needed because the low concentrations of initiating groups cannot generate a sufficient concentration of deactivator, or persistent radical, in the contacting solution to provide a controlled grafting from reaction. Alternatively, a certain amount of CuII species can be added to the contacting monomer solution at the beginning of the reaction to achieve persistent radical effect.(5) Tapping mode AFM images of pDMAEMA grafts prepared by grafting from a modified Si-OH surface imaged in air under ambient conditions are shown below in images a-c. Images of surfaces with tethered grafts formed with higher than 20% initiator coverage revealed the presence of dense, relatively smooth films, with occasional patchy defects. At lower initiator coverage, the tethered grafts were increasingly patchy, and at 1% coverage and below, they appeared to be highly uniform, isolated patches.

AFM images of grafts grown from the Si-H substrates are shown in images d-f. It appears that, in comparison with oxidized silicon substrates with the same assumed initiator coverage, films grown from Si-H substrates were denser, showing fragmentation into patches only at the lowest initiator coverage (Image f). The use of either substrate (Si-OH vs. Si-H) seems to yield equally uniform graft layers when the concentration of initiating group on the surface is ≥20%.
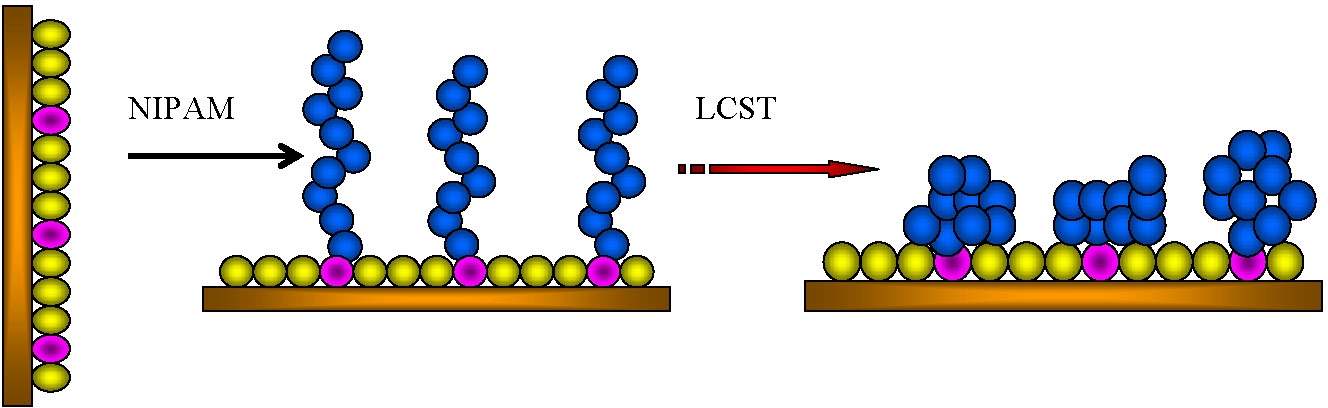
When a surface tethered poly(N-isopropylacrylamide) was synthesized, by surface initiated polymerization, the hydrophobicity of the resulting surface changed as the temperature was increased through the LSCT. However in contrast to "free" poly(N-isopropylacrylamide) the surface tethered polymer does not respond to external stimuli within seconds. The following images show an air bubble in water in contact with the modified surface at different temperatures. The contact angle increased from 62 degrees to 79 degrees as the tethered NIPAM chains were transformed at the LCST.

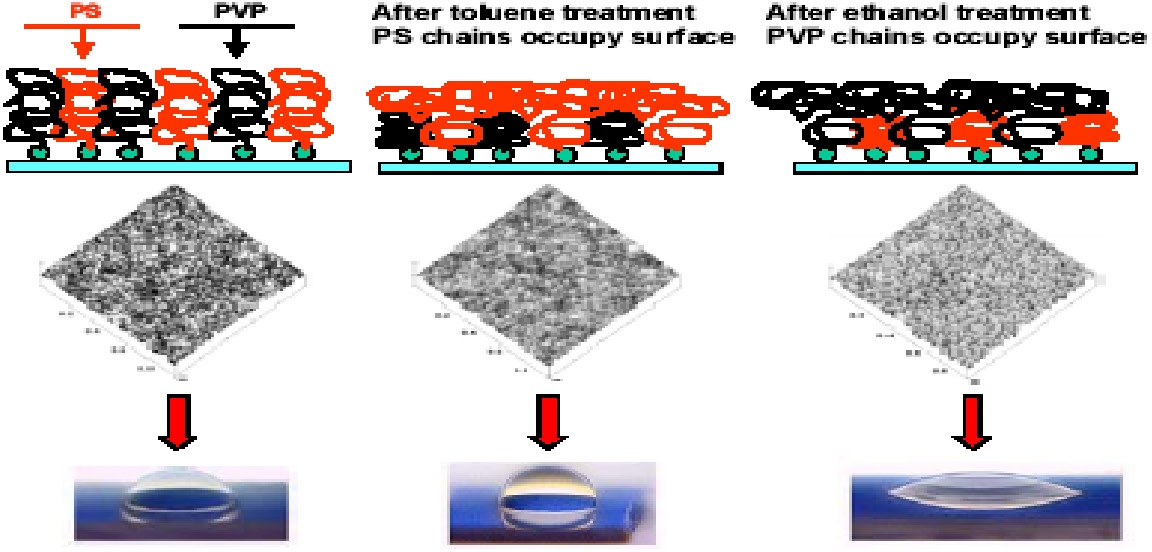
The following series of images are taken from reference 4 and shows schematic representations of a surface modified with dual responsive tethered polymers. The fundamental chemical nature of the underlying material becomes hidden by the presence of a film only a few Angstroms thick. The interaction of the whole system with the surrounding environment is governed by the tethered polymeric coatings. Scanning Probe Microscopy topography images and wetability experiments for hybrid polymer layers consisted of polystyrene and poly(vinyl pyridine) show how the response of the surface is modified by prior environmental challenges, undergoing reversible changes from a hydrophobic surface to a more hydrophilic surface.
REFERENCES
(1) Matyjaszewski, K.; Dong, H.; Jakubowski, W.; Pietrasik, J.; Kusumo, A. Langmuir 2007, 23, 4528-4531.
(2) Pyun, J.; Kowalewski, T.; Matyjaszewski, K. Polymer Brushes 2004, 51-68.
(3) Pyun, J.; Jia, S.; Kowalewski, T.; Matyjaszewski, K. Macromolecular Chemistry and Physics 2004, 205, 411-417.
(4) Husseman, M.; Malmstroem, E. E.; McNamara, M.; Mate, M.; Mecerreyes, D.; Benoit, D. G.; Hedrick, J. L.; Mansky, P.; Huang, E.; Russell, T. P.; Hawker, C. J. Macromolecules 1999, 32, 1424-1431.
(5) Matyjaszewski, K.; Miller, P. J.; Shukla, N.; Immaraporn, B.; Gelman, A.; Luokala, B. B.; Siclovan, T. M.; Kickelbick, G.; Vallant, T.; Hoffmann, H.; Pakula, T. Macromolecules 1999, 32, 8716-8724.
