Catalyst Reduction
ARGET and ICAR
Biphasic Systems
-
A Hybrid Catalyst System
-
A “Soluble” Hybrid Catalyst System
-
Mixed Solvent Polymerization
-
Liquid/Liquid Biphasic Polymerization
ARGET and ICAR:
As shown in the figure displaying relative catalyst activity for copper based catalysts at the end of the page on Catalyst Development(1) catalysts have been developed that show a broad range of activity. It is possible, therefore, to select an active catalyst and run the reaction with lower levels of catalyst.(2,3) In the page on "Procedures for Initiation of an ATRP Reaction" we cover much of the background on the development procedures that allow conducting an ATRP with low catalyst concentration. Indeed with the extension of the concept of AGET ATRP(4,5) to include continuous regeneration of the transition metal complex throughout the reaction, ARGET ATRP,(3-9) it is possible to reduce the level of catalyst below that of natural termination reactions and ATRP can be conducted with ppm levels of catalyst.
ARGET ATRP arose when we considered the implications of the convenient procedure for initiating an ATRP system using high activity catalyst complexes described in AGET ATRP, where the activators are generated by electron transfer (AGET) ATRP. We realized it should be possible to use the reducing agents (e.g. tinII 2-ethylhexanoate, glucose, ascorbic acid, amines, Cu0 etc. for ARGET or AIBN for ICAR) to constantly regenerate the ATRP activator, the CuI species, from the CuII species formed during termination processes, without directly or indirectly producing initiating species that generate new chains.(4) A detailed examination of the ATRP rate law (Rp) shows that the polymerization rate depends only on the ratio of the concentration of [CuI] to [X-CuII], and does NOT depend on the absolute concentration of the copper complexes, therefore in principle, one could reduce the absolute amount of copper complex to ppm levels without affecting the polymerization rate.
![]()
However, a residual amount of deactivating species (i.e. X-CuII) is required for a well-controlled polymerization since both, molecular weight distribution and initial molecular weight, depend on the ratio of the propagation and deactivation rate constants and the concentration of deactivator.
![]()
This means that in order to obtain polystyrene with Mw/Mn~1.2, when targeting a DP~200 and 90% conversion at ~100 0C, the actual amount of X-CuII species required to conduct a controlled reaction is ~2 ppm (kp~103 M-1s-1 and kda~107 M-1s-1), meaning that the concentration of the X-CuII species could be reduced over 1,000 times from the level typically used in the initial ATRP's. Unfortunately, if the amount of CuI is reduced 1,000 fold, unavoidable radical-radical termination reactions irreversibly consume all of the activators present in the reaction media and the reaction slows down or stops; i.e. if ~10% of chains terminate and the amount of CuI initially added to the system was below 10 mole% of the initiator all CuI would be consumed. It was recognized that this situation could be overcome if there was constant regeneration of the CuI activator species by environmentally acceptable reducing agents to compensate for any loss of CuI by termination.
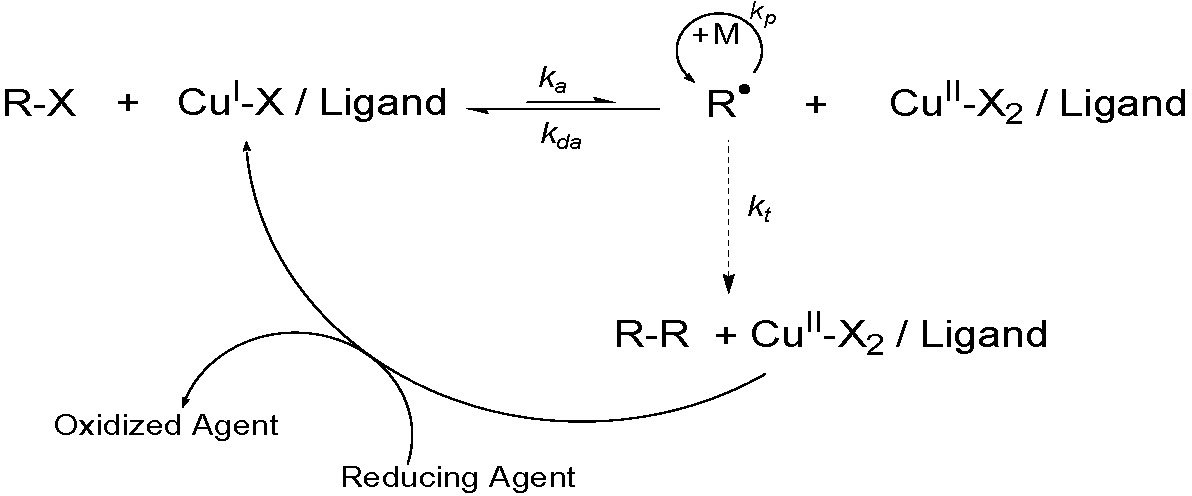
Generally, it is desirable to add an excess of the ligands compared to the transition metal complex, in order to compensate for competitive complexation by monomer/solvent/reducing agent all present in significant molar excess compared to the transition metal.(5-8) Recently, the role of excess ligand, or ligand substitute, has been expanded to include its potential as a reducing agent.(10,11)
For example, styrene was polymerized by the addition of 5 ppm of CuCl2/Me6TREN and 500 ppm of Sn(EH)2 to the reaction resulting preparation of a polystyrene with Mn = 12,500 (Mn,th = 12,600) and Mw/Mn = 1.28.
ARGET and ICAR ATRP systems can be also used for successful chain extension of polyacrylate and polystyrene macroinitiators with methacrylates. About 50 ppm of copper was applied to polymerization of macroinitiators and during chain extension. 10 mol. % of styrene was introduced to the system to overcome the problem associated with inefficient initiation from the added macroininiator at the first extension step from polyacrylates/styrenes by MMA.(12)
Reducing the concentration of copper catalyst present in the reaction below 1-5 ppm resulted in preparation of polymers with higher PDI (broader MWD). However, chain end functionality is not affected, so polydisperse macroinitiators can be successfully utilized for chain extension forming broad-narrow diblock copolymers. This novel and robust method allows creating (stabilizing) interesting morphologies (e.g. hexagonally perforated lamellae or highly asymmetric lamellae) in custom synthesized polymeric materials.(13)
An added advantage of using low levels of catalyst in an ATRP is that catalyst induced side reactions are reduced and it is possible to prepare higher molecular weight copolymers(9,14) and conduct the reaction in the presence of limited amounts of oxygen.(15)
REFERENCES
(1) Honigfort, M. E.; Brittain, W. J. Macromolecules 2003, 36, 3111-3114.
(2) Min, K.; Gao, H.; Matyjaszewski, K. Journal of the American Chemical Society 2005, 127, 3825-3830.
(3) Jakubowski, W.; Matyjaszewski, K. Macromolecules 2005, 38, 4139-4146.
(4) Matyjaszewski, K.; Bombalski, L.; Jakubowski, W.; Min, K.; Spanswick, J.; Tsarevsky, N. In PCT Int. Appl.; (Carnegie Mellon University, USA). WO 2005087819, 2005; p 96 pp.
(5) Jakubowski, W.; Min, K.; Matyjaszewski, K. Macromolecules 2006, 39, 39-45.
(6) Jakubowski, W.; Matyjaszewski, K. Angewandte Chemie, International Edition 2006, 45, 4482-4486.
(7) Matyjaszewski, K.; Jakubowski, W.; Min, K.; Tang, W.; Huang, J.; Braunecker, W. A.; Tsarevsky, N. V. Proceedings of the National Academy of Sciences 2006, 103, 15309-15314.
(8) Min, K.; Gao, H.; Matyjaszewski, K. Macromolecules 2007, 40, 1789-1791.
(9) Pietrasik, J.; Dong, H.; Matyjaszewski, K. Macromolecules 2006, 39, 6384-6390.
(10) Kwak, Y.; Matyjaszewski, K. Polymer International 2009, 58, 242-247.
(11) Tang, H.; Shen, Y.; Li, B.-G.; Radosz, M. Macromol. Rapid Commun. 2008, 29, 1834-1838.
(12) Mueller, L.; Jakubowski, W.; Tang, W.; Matyjaszewski, K. Macromolecules 2007, 40, 6464-6472.
(13) Listak, J.; Jakubowski, W.; Mueller, L.; Plichta, A.; Matyjaszewski, K.; Bockstaller, M. R. Macromolecules 2008, 41, 5919-5927.
(14) Dong, H.; Tang, W.; Matyjaszewski, K. Macromolecules 2007, 40, 2974-2977.
(15) Matyjaszewski, K.; Dong, H.; Jakubowski, W.; Pietrasik, J.; Kusumo, A. Langmuir 2007, 23, 4528-4531.
