Materials You Can Prepare
Gradient Copolymers
Block Copolymers
-
Poly(styrene-b-t-butyl acrylate)
-
Poly(4-VP)-b-P(MMA)
-
PODMA-b-PMMA
-
PnBA-b-PS by ARGET ATRP
-
PolyBA-b-Poly(MMA-co-St) and PolySt-b-Poly(MMA-co-St) Using ARGET or ICAR ATRP
-
PolyBA-b-Poly(MMA-co-St) by ICAR ATRP
-
Polymers via ARGET ATRP
-
AGET ATRP of BA in miniemulsion with PEO-PS-Br as macroinitiator and stabilizer.
Gradient Copolymers
A One-pot Forced Gradient Copolymerization of nBA and tBA by AGET ATRP in Miniemulsion
(Min, K.; Oh, J. K.; Matyjaszewski, K. Journal of Polymer Science, Part A: Polymer Chemistry 2007, 45, 1413-1423.)
Basic Conditions:
The monomers (nBA (1.8 g, 14 mmol), tBA (1.8 g, 14 mmol)), catalysts (CuBr2 (0.0125 g, 0.056 mmol) and BPMODA (0.0254 g, 0.056 mmol)), mono-functional initiator EBriB (21µL, 0.14 mmol) and co-stabilizer hexadecane (0.17 mL) were mixed in a round bottom flask at 60 ºC. After the formation of the Cu(II) complex, an aqueous Brij 98 solution (16 mL, 5 mol/L) was added to the system. The resulting mixture was subject to sonication (Heat Systems Ultrasonics W-385 sonicator; output control set at 8 and duty cycle at 70%) for 1 min in an ice bath (to prevent a possible temperature rise resulting from sonication) to form a stable miniemulsion. The stable miniemulsion was transferred to a Schlenk flask and bubbled with nitrogen for 30 min. The flask was immersed into an oil bath preheated at 80 ºC. The polymerization was initiated by the injection of a pre-deoxygenated aqueous solution (0.5 mL) of ascorbic acid (0.0044 g) to activate the copper complex. Aliquots were withdrawn from the reaction at the different time intervals to determine conversion by Gas Chromatography (GC). The samples were dried under vacuum and dissolved in THF before they were subjected to gel permeation chromatography (GPC) for molecular weight analysis. The polymerizations were stopped by exposing the catalyst to air.
Examples of forced gradient copolymerization by AGET ATRP in miniemulsion
Several monomer pairs were subject to the preparation of forced gradient copolymers by AGET ATRP in miniemulsion. In a typical run, CuBr2 (0.0125 g, 0.056 mmol), BPMODA (0.0254 g, 0.056 mmol), nBA (1.8 g, 14 mmol), EBriB (21 µL, 0.14 mmol) and hexadecane (0.17 mL) were mixed in a 25 mL round bottom flask at 60 oC. After the formation of the CuII complex, an aqueous solution of Brij 98 (16mL, 5 mmol/L) was added to the system and the mixture was subject to sonication for 1 min in an ice bath to form a stable miniemulsion. The stable miniemulsion was transferred to a Schlenk flask and bubbled with nitrogen for 30 min. The flask was immersed in an oil bath preheated at 80 oC. The polymerization was initiated by injection of a pre-deoxygenated aqueous solution (0.5 mL) of ascorbic acid (0.0044 g). Simultaneously the pre-deoxygenated tBA monomer (1.8 g, 14 mmol) was continuously added to the miniemulsion at a predetermined rate with the aid of a syringe pump (KdScientific KDS 210 infusion/withdrawal syringe pump). Aliquots were periodically withdrawn via a pre-degassed syringe to monitor monomer conversion by gravimetry and 1H NMR. The samples were dried under vacuum and dissolved in THF before they were subjected to GPC for molecular weight analysis. The polymerizations were stopped by exposing catalysts to air. In the present study, the following monomer pairs: nBA/tBA, BMA/MMA and nBA/St, were selected for preparation of forced gradient copolymers in miniemulsion. The polymerization conditions are summarized in the following table.
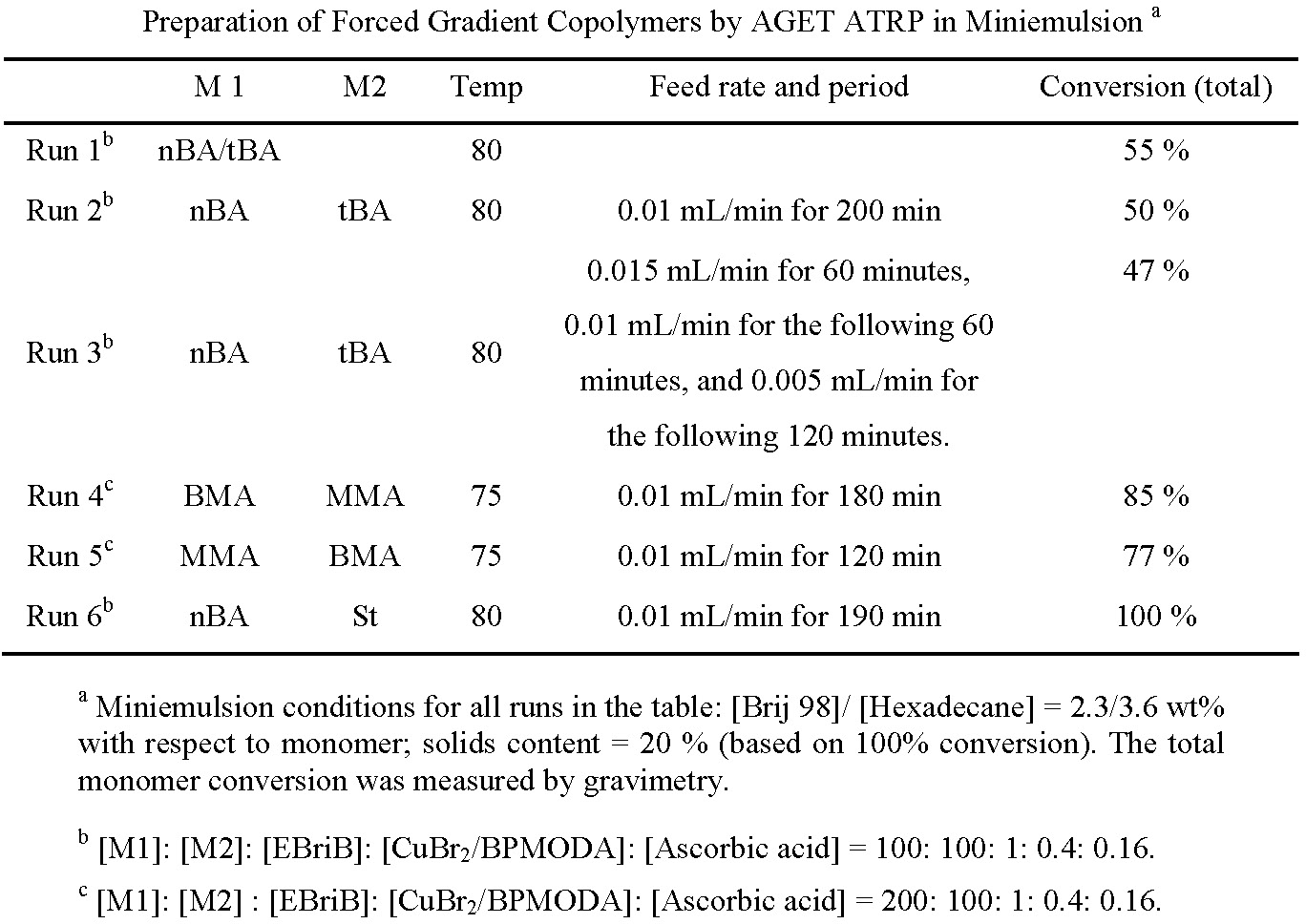
Cumulative compositions (A) and instantaneous compositions (B) of nBA and tBA in the forced gradient copolymer resulting from AGET ATRP in miniemulsion with tBA as a fed monomer and the feeding rate of 0.01 mL/min for 200 minutes are shown in the following figures. Polymerization conditions: Run 2 in above table.
Block copolymers
(Beers, K. L.; Woodworth, B.; Matyjaszewski, K. J. Chem. Educ. 2001, 78, 544-547. Matyjaszewski, K.; Beers, K. L.; Woodworth, B.; Metzner, Z. J. Chem. Educ. 2001, 78, 547-550. Matyjaszewski, K.; Qiu, J.; Tsarevsky, N. V.; Charleux, B. J. Polym. Sci., Part A: Polym. Chem. 2000, 38, 4724-4734.)
The best order of chain extension from (macro)initiators: AN > MMA > MA > Sty.
Going from MA to MMA or AN requires halogen exchange (CuCl-based catalyst). In chain extension of Sty with MMA or AN, halogen exchange improves control but not sufficiently.
It is also possible to prepare block copolymers where one or more segments of the block copolymer had been prepared by a non-CRP procedure. The only requirement is to ensure the terminal functional groups present on the initial functional polymer can be converted into radically transferable atom(s) for the second controlled ATRP step.
(Coca, S.; Matyjaszewski, K. Macromolecules 1997, 30, 2808-2810. Matyjaszewski, K.; Coca, S.; Gaynor, S. G.; Nakagawa, Y.; Jo, S. M. In PCT Int. Appl.; (Carnegie-Mellon University, USA). WO 9801480, 1998; p 105 pp. Bernaerts, K. V.; Schacht, E. H.; Goethals, E. J.; Du Prez, F. E. Journal of Polymer Science, Part A: Polymer Chemistry 2003, 41, 3206-3217.)
Synthesis of Diblock Copolymer Poly(styrene-b-t-butyl acrylate)
(Matyjaszewski, K.; Qiu, J.; Tsarevsky, N. V.; Charleux, B. J. Polym. Sci., Part A: Polym. Chem. 2000, 38, 4724-4734.)
There are a series of chain extensions described in references 2-4, one of which is excerpted below.
All polymerization reactions were catalyzed by CuBr/PMDETA. The GPC traces showed relatively clean block chain extension, with movement of the entire polymer distribution to higher molecular weights. This confirmed that the end functionality was maintained throughout the reactions. CuBr2 was added to the reactions where lower molecular weight p(tBA) blocks were targeted. On the basis of other results from this laboratory the addition of CuII can slow down the rate of the reaction and increase the efficiency of initiation; i.e. reduce termination reactions required to form the ATRP equivalent of the persisitent radical, the deactivator. In addition, the use of a more polar solvent, such as acetone, can also result in a more controlled reaction through better solvation of both the activator and deactivator. Both of these modifications help to produce polymers with lower polydispersities than those prepared without either the added CuII or the polar solvent. For higher molecular weight p(tBA) blocks, no additional CuII was necessary because decreasing the initiator and catalyst concentrations by dilution functioned in the same manner.
Typical run conditions:
The macroinitiator and CuBr were initially added to the flask. The flask was sealed with a rubber septum, degassed, backfilled three times with N2, and then left under N2. The monomer and internal standard/solvent were added, and only after dissolution of the macroinitiator was the PMDETA ligand added. An initial sample was removed to measure the initial ratio of monomer and internal standard for comparison against the final ratio. Reactions were run in a thermostated oil bath for the desired amount of time, after which time the flask was removed, the final monomer conversion was measured, and the molecular weight analyses were performed.
The most important aspect of these polymerizations was the optimization of the catalyst system to obtain the desired molecular weights, while at the same time maintaining reasonable rates of polymerization, resulting in polymers with narrow molecular weight distributions.
[tBA]:[CuI]:[CuII]:[ PMDETA] = 50:1:0.5:0.025:0.525 plus 25% acetone at 60°C for 180 minutes provided 43% conversion and a Mn, th of 3,800 compared to Mn, exp of 3,725.
(Mi, H.; Zhao, Z.; Wang, C.; Long, Y. Proceedings of International Forum on Green Chemical Science & Engineering and Process Systems Engineering, Tianjin, China, Oct. 8-10, 2006 2006, 1, 554-558.)
Poly(4-VP)-b-P(MMA)
A dry glass tube with a stir bar was charged with CuCl (2.6 mg), the PMMA-Cl macroinitiator (Mn, SEC = 7700, Mw/Mn = 1.06, 100 mg), DMF (0.5 mL), Me6TREN (7.1 μL), and 4VP (1.0 mL). Three freeze-pump-thaw cycles were performed, and the tube was sealed under vacuum and placed in an oil bath held at 50 °C. After 3.0 h, the tube was opened, and the contents were dissolved in DMF. Monomer conversion and SEC measurements were performed on the reaction mixture.
For the 1H NMR analysis, the block copolymer was recovered by removing the solvent under reduced pressure, redissolving the polymer in chloroform, passing the polymer solution through an alumina column to remove the metal containing residues, and precipitation in cold hexane. The polymer was dried at 80 °C under vacuum for 48 h. 1H NMR indicated that the block copolymer had 87 wt % of P4VP and a molecular weight of Mn,NMR = 62,500 which was in good agreement with the calculated value (Mn,Cal = 63,800 at 84% monomer conversion) assuming quantitative cross-propagation and the absence of any chain-breaking reactions. However, the molecular weight determined by SEC with PMMA standards was much higher (Mn,SEC = 89,500).
Synthesis of a PODMA-b-PMMA Diblock Copolymer by AGET ATRP
The PODMA macroinitiator (Mw=13800, Mw/Mn =1.10) (3.0g, 0.2 mmol), CuCl2 (31 mg, 7.3 x 10-5 mol) and monomer (MMA, 9.4 g, 0.1 mol) were were added to a 25 mL Schlenk flask and bubbled with nitrogen for 15 minutes forming a clear solution. A purged solution of PMDETA (48 µl, 0.23 mmol) in toluene (5ml) was added, and the mixture was stirred. A solution of Sn(EH)2 (34 μl, 0.1 mmol) in toluene (4ml) was added next, and an initial sample was taken. The flask was placed in a thermostated oil bath at 90 °C and stirred. The polymerization was stopped after 17 hours by opening the flask and exposing the catalyst to air.
[MMA]:[MI]:[CuCl2]:[Sn(EH)2]:[PMDETA] = 500:1:0.073:0.5:2.3
Conversion = 58%,Mn,ex 38100; Mw/Mn 1.09.
Synthesis of Diblock Copolymer PnBA-b-PS by ARGET ATRP
A PnBA macroinitiator (Mw=19400, Mw/Mn =1.26) (2.35 g, 0.12 mmol) was dissolved in styrene monomer (St, 2.75 ml, 24.0 mol) in a 25 mL Schlenk flask and bubbled with nitrogen for 15 minutes. Next, a solution of CuCl2 (4.84 x 10-2 mg, 0.36 x 10-3 mmol)/Me6TREN (0.10 ml, 0.36 x 10-3 mmol) complex in degassed anisole (0.7 ml) was added. The resulting mixture was stirred for 10 minutes and then 3.9 ml (1.2 x 10-2 mmol) of a purged solution of Sn(EH)2 and Me6TREN (3.2 ml, 1.2 x 10-2 mmol) in anisole (0.7 ml) was added. An initial sample was taken and the sealed flask was placed in thermostated oil bath at 110 oC. Samples were taken at timed intervals and analyzed by GC and GPC. The polymerization was stopped after 30.8 h by opening the flask and exposing the catalyst to air. Mn, GPC = 34,900 and Mw/Mn = 1.18, conversion = 88%.
Synthesis of PolyBA-b-Poly(MMA-co-St) and PolySt-b-Poly(MMA-co-St) Using ARGET or ICAR ATRP
(Mueller, L.; Jakubowski, W.; Tang, W.; Matyjaszewski, K. Macromolecules 2007, 40, 6464-6472.)
As noted in the introduction to this synthesis section chain extension from a secondary macroinitiator to controlled polymerization of a tertiary monomer is not efficient and "halogen exchange" was developed. However halogen exchange can not work when low concentrations of catalyst are employed and another means of control had to be developed. This was conducting a copolymerization in the second block.
PolyBA-b-Poly(MMA-co-St):
PolyBA-Br (1 g, 0.0556 mmol) and anisole (1 mL) were added to a Schlenk flask, and purged with nitrogen. Next, a mixture of MMA (1.1 mL, 10.0 mmol) and St (0.13 mL, 1.11 mmol) that had been purged with nitrogen was added to the flask, followed by a olution of CuCl2 (0.075 mg, 0.556 μmol) and TPMA (0.97 mg, 3.33 μmol) in purged anisole (0.5 mL). Sn(EH)2 (1.8 μL, 5.56 μmol) in purged anisole (0.5 mL) was added to activate a fraction of the catalyst complex and begin the polymerization. An initial sample was taken via purged syringe, and the sealed flask was placed in an oil bath at 90 °C. Samples were taken at timed intervals and analyzed by gas chromatography (GC) and gel-permeation chromatography (GPC) to follow the progress of the reaction. The polymerization was stopped by opening the flask and exposing the catalyst to air, and a polyBA-b-poly(MMA-co-St) block copolymer was obtained.
When a PolyBA-Br macroinitiator of Mn = 18,000 and Mw/Mn = 1.12 was chain extended with MMA/St/PBA-Br/CuCl2/Sn(EH)2 = 180/20/1.0/0/01/0.06/0.1 a block copolymer with Mn = 30,100 and Mw/Mn 1.23 was obtained at 83% monomer conversion.
PolySt-b-Poly(MMA-co-St):
A similar procedure was used to synthesize polySt-b-poly(MMA-co-St). A polystyrene macroinitiator of Mn = 19,500 and Mw/Mn = 1.09 was chain extended to provide a block copolymer .
Conversion 86% Mn = 38,100 and Mw/Mn = 1.30.
Synthesis of PolyBA-b-Poly(MMA-co-St) by Initiators for Continuous Activator Regeneration (ICAR) ATRP
In this case 5.56 μmol of AIBN was added to the reaction in place of Sn(EH)2, 0.556 μmol of TPMA was used as lignd, and the oil bath was thermostated at 60 °C. Under these sircumstances when a PolyBA-Br macroinitiator of Mn = 18,000 and Mw/Mn = 1.12 was chain extended with MMA/St/PBA-Br/CuCl2/AIBN = 180/20/1.0/0/01/0.01/0.1 a block copolymer with Mn =36,000 and Mw/Mn 1.38 was obtained.
Synthesis of Polymers via ARGET ATRP by Using Ascorbic Acid as Reducing Agent
(Min, K.; Gao, H.; Matyjaszewski, K. Macromolecules 2007, 40, 1789-1791.)
The limited solubility of ascorbic acid in organic solvents, such as anisole, provides a slow reduction of Cu(II) species which facilitates well-controlled atom transfer radical polymerization (ATRP) of various monomers using the recently developed activators regenerated by electron transfer (ARGET) technique.
In polymerizations with 25 ppm, or less than 25 ppm of Cu, a stock solution of Cu(II)/Me6TREN was made by dissolving CuBr2 (0.0223 g, 0.1 mmol), Me6TREN (264 μL, 1 mmol) in 5 mL of DMF. In a typical run, EBriB (14.7 μL, 0.1 mmol), MA (3.6 mL, 40 mmol), 50 μL of the Cu(II) stock solution and anisole (2.4 mL) were added to a 10 mL Schlenk flask. The flask was sealed and then deoxygenated by several freeze-pump-thaw cycles. Then ascorbic acid (0.0088 g, 0.05 mmol) was added to the flask and the flask was immersed in an oil bath, thermostated at 60 oC, to initiate polymerization. Aliquots were taken at intervals to measure conversion and to examine the evolution of molecular weight.
Synthesis of poly(methyl acrylate) (PMA) by ARGET ATRP using ascorbic acid as reducing agent.
Polymerization temperature: 60 oC. Polymerizations were carried out in 60% v/v solutions. Initiator: ethyl 2-bromoisobutyrate (EBriB). Ligand: Me6TREN. The amount of ligand has been fixed at 10 times the amount of Cu(II) initially added to the reaction in order to assure coordination of the ligand with Cu(II) in the presence of a large excess of monomer.
When the concentration of Cu: molar ratio of Cu to monomer 25 ppm and [MA]: [I]: [Cu(II)]= 400: 1: 0.01; conversion reached 87% after 5.3 hours and Mn (theo) =30,000 Mn (GPC) = 27,700 and Mw/Mn 1.19.
Continuing the reaction to 98% conversion provided a polymer with Mn (theo) =33,750 Mn (GPC) = 34,400 and Mw/Mn 1.18.
When the concentration of copper was reduced to 10 ppm and [MA]: [I]: [Cu(II)]= 400: 1: 0.004 the reaction reached 91% conversion provided a polymer with Mn (theo) =31,340 Mn (GPC) = 29,700 and Mw/Mn 1.40.
All polymerizations carried out with 25 ppm or lower concentration of Cu resulted in the preparation of essentially colorless polymers; therefore, catalyst removal may not be necessary for some applications.
Synthesis of poly(butyl acrylate), poly(methyl methacrylate) and polystyrene by ARGET ATRP with ascorbic acid as reducing agent*
|
Entry |
M |
Ascorbic acid (to Cu) |
Cu (ppm)a |
Solvent |
Time (h) |
Conv. |
Mn(theo) |
Mn(GPC) |
Mw/Mn |
|
7 b |
BA |
10 |
25 |
Anisole |
21.5 |
89% |
45500 |
43000 |
1.28 |
|
8 b |
MMA |
10 |
25 |
Anisole |
18 |
59% |
23260 |
30700 |
1.27 |
|
9 c |
St |
10 |
25 |
Anisole |
64.7 |
68% |
27580 |
25200 |
1.18 |
|
10 d |
St |
10 |
25 |
Anisole |
52 |
55% |
53540 |
60900 |
1.27 |
* Polymerizations were carried out in 60 % v/v solutions.
Initiator: EBriB for BA and St, ethyl α-bromophenylacetate for MMA. Ligand: Me6TREN.
The amount of ligand has been fixed at 10 times the amount of Cu(II) initially added to the reaction.
a Concentration of Cu: molar ratio of Cu to monomer.
b [M]: [I]: [Cu(II)]= 400: 1: 0.01. Reaction temperature: 60 oC; c [M]: [I]: [Cu(II)]= 400: 1: 0.01.
Reaction temperature: 90 oC; d [St]: [PMA-Br]: [Cu(II)]= 400: 1: 0.01. Reaction temperature: 90 oC.
AGET ATRP of BA in miniemulsion with PEO-PS-Br as macroinitiator and stabilizer.
(Li, W.; Min, K.; Matyjaszewski, K.; Stoffelbach, F.; Charleux, B. Macromolecules 2008, 41, 6387-6392.)
PEO-PS-Br block copolymers with various molecular weights (PEO44-PS17-Br, PEO120-PS29-Br and PEO120-PS15-Br) were used as reactive surfactants for AGET ATRP of BA in miniemulsion. Typically, CuBr2 (0.0012 g, 0.005 mmol) and BPMODA (0.0024 g, 0.005 mmol) are dissolved in BA (0.84 mL, 5.87 mmol) in a round bottom flask at 60 °C for 1 h. The resulting solution was then cooled to room temperature and hexadecane (0.027 g) was dissolved in it. PEO120-PS15-Br (0.1 g, 0.013 mmol initiating sites) was dispersed in 8 g water. The aqueous PEO-PS-Br solution was then added to the organic solution. The resulting mixture was subjected to sonication in an ice bath (Heat Systems Ultrasonics W-385 sonicator; output control set at 8 and duty cycle at 70% for 5 min). The resulting stable miniemulsion was transferred to a Schlenk flask and purged with nitrogen for 30 min before being immersed in an oil bath thermostated at 70 °C. A predeoxygenated aqueous solution of ascorbic acid was injected into the miniemulsion to initiate the polymerization.
AGET ATRP of BA in miniemulsion with PEO-PS-Br as macroinitiator and stabilizer and EBiB as co-initiator.
EBiB was used as co-initiator with PEO-PS-Br macroinitiator. The procedure was similar to that described above. Typically, before conducting a miniemulsion polymerization, CuBr2 (0.0095 g, 0.043 mmol) and BPMODA (0.0192 g, 0.043 mmol) were dissolved in BA (2.45 mL, 17.1 mmol) in a round bottom flask at 60 °C for 1 h. The resulting solution was then cooled to room temperature and hexadecane costabilizer (0.079 g), and EBiB initiator (14 µL, 0.096 mmol), were added. PEO120-PS15-Br (0.08 g, 0.011 mmol initiating sites) was dispersed in 15 g water. The aqueous PEO120-PS15-Br solution was then added to the organic solution. The resulting mixture was sonicated for 5 min. The resulting stable miniemulsion was transferred to a Schlenk flask and purged with nitrogen for 30 min before being immersed in an oil bath thermostated at 70 °C. A predeoxygenated aqueous solution of ascorbic acid was injected into the miniemulsion to activate the catalyst and initiate the polymerization. Samples were taken at timed intervals to measure the conversion gravimetrically and to determine the molecular weight by GPC.
PEO44-Br homopolymer was also used as reactive surfactant for AGET ATRP of BA in minemulsion either with or without EBiB as a co-initiator using similar procedures.
