Brooke M. McCartney
Associate Professor
Address:
600D Mellon Institute
Department of Biological Sciences
Carnegie Mellon University
4400 Fifth Avenue
Pittsburgh, PA 15213
Phone: 412-268-5195 (Office)
412-268-9820 (Lab)
Fax: 412-268-7129

Education
Ph.D., Duke University
Postdoctoral Appointment, University of North Carolina at Chapel Hill
Research
The McCartney lab investigates fundamental cellular mechanisms that drive developmental processes including oogenesis and systemic growth in Drosophila.
In oogenesis, we are dissecting the mechanisms controlling the assembly, organization, dynamics and function of two co-aligning and co-regulating cytoskeletal networks: microtubules (MTs) and actin filaments bundled into cables. A significant gap in our understanding of the cytoskeleton is how different cytoskeletal networks interact and coregulate. The actin cables are produced by fifteen nurse cells connected to each other and to the oocyte through ring canals. Late in oogenesis (s11), the nurse cell network rapidly expels its cytoplasm into the oocyte through ring canals (“dumping”). Immediately preceding this step (s10B), actin filaments initiate at the cell cortex through the activity of actin assembly factors, are bundled into cables by bundling proteins and elongate toward the nucleus in each nurse cell. The growing actin cables contact the nuclei and push them away from ring canals, preventing obstruction during cytoplasmic dumping. Our current work focuses on the crosstalk between actin cable arrays and MT networks. We discovered that the uncharacterized, acentrosomal, acetylated and stable MT network is necessary for cable initiation and for promoting the normal cable elongation rate. Perturbing actin cable assembly directly by interfering with filament production or bundling produced distinct effects on the MT network.
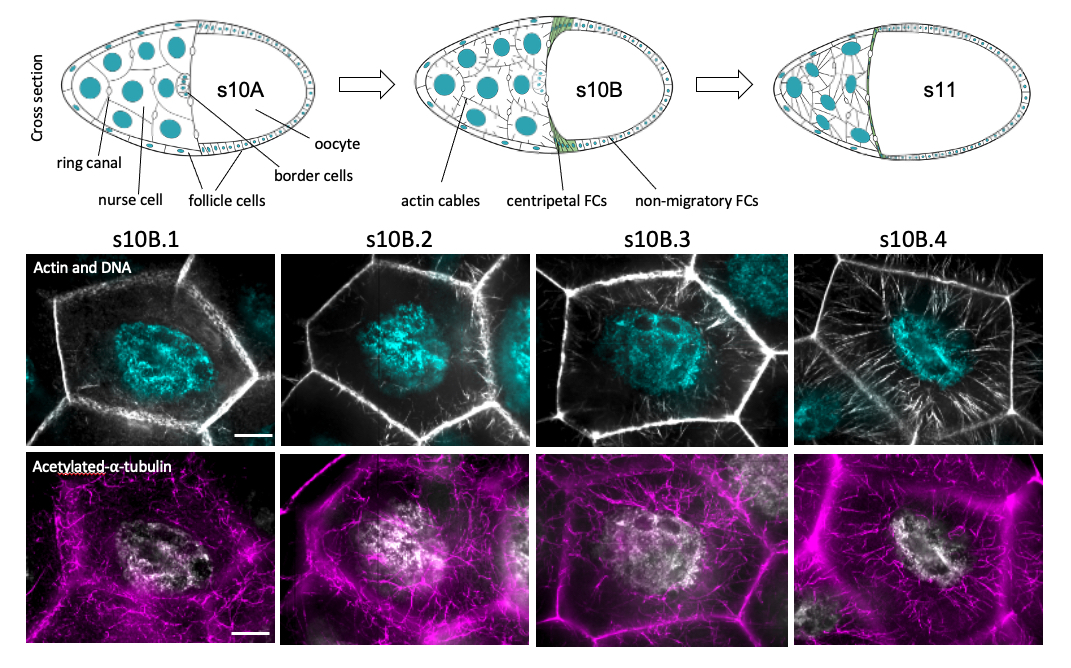
Stable actin structures like microvilli and stereocilia exhibit dynamic maintenance, the balanced addition and subtraction of monomers in established filaments. Surprisingly, we found that both microtubules and actin filament dynamic maintenance are required to maintain the cortical continuity of the growing actin cables. Further, our data suggest a novel regulation of dynamic maintenance by microtubules. We will be pursuing these questions in the future.
Organismal (systemic) growth is a key feature of animal development. Although genes and diet play integral roles in controlling growth, exciting recent advances have revealed important roles for the bacterial gut microbiota. While a connection between the microbiota and animal growth has been established, there are many gaps in our understanding of the underlying molecular mechanisms. We discovered an unexpected, novel collaboration between Drosophila Arc1 and bacteria of the gut microbiota to promote systemic growth. Recent surprising work demonstrated that mammalian Arc (mArc) and fly Arc1 proteins self-assemble into viral capsid-like structures that transport mArc/Arc1 mRNA and other RNAs via extracellular vesicles (EVs) across synapses and between cells, constituting a novel mechanism of intercellular communication. Loss of the microbiota in wild type Drosophila results in a prolonged growth period that produces smaller animals.
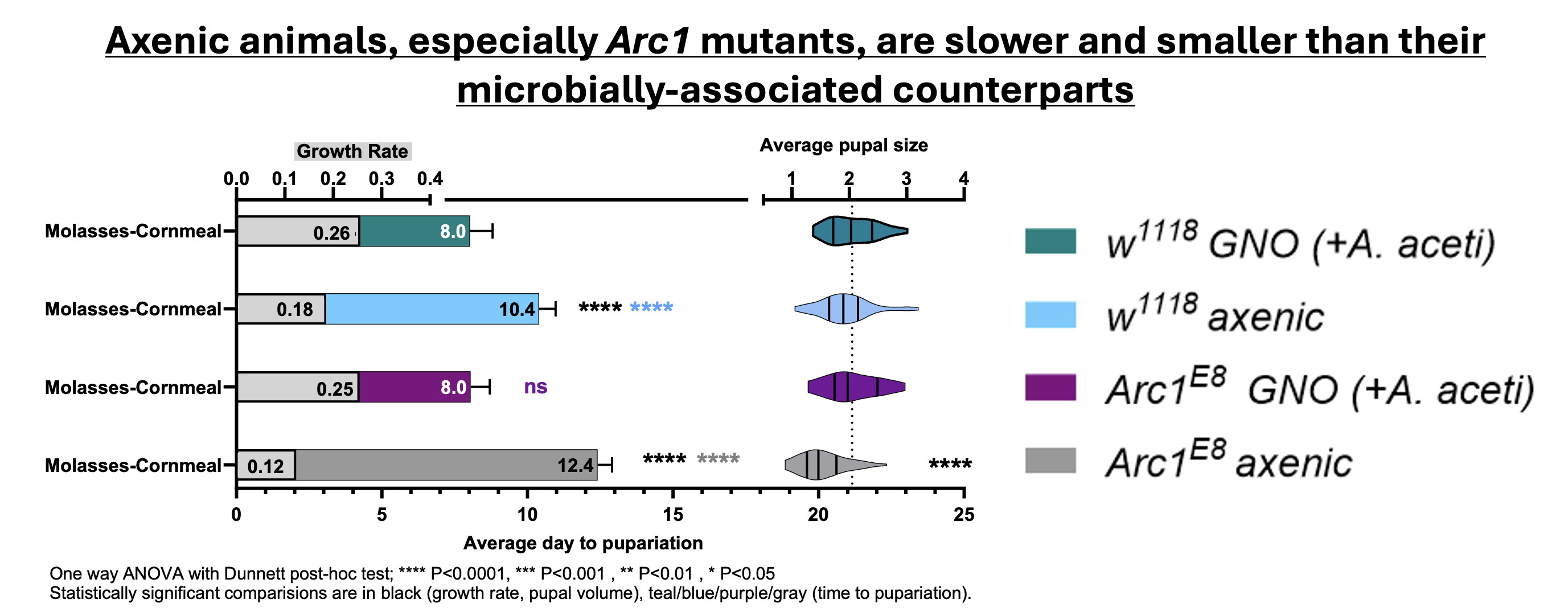
We found that while bacteria-associated Arc1 mutants exhibit wild type growth, loss of Arc1 in germ-free animals dramatically enhanced this microbe-dependent growth defect and significantly dysregulated Insulin/Insulin-like growth factor signaling. Monoassociating Arc1 mutants with Acetobacter aceti, isolated from our lab fly microbiota, could rescue growth defects in Arc1 mutants. We are currently working to identify the specific molecular contributions that Arc1 and A. aceti make to systemic and proportional growth regulation and the role of diet in this interaction.
Publications
Recent notable publications:
Developmentally regulated actin-microtubule crosstalk in Drosophila oogenesis
Chou, W-C, M. Lakonishok, W. Lu, V. Gelfand and B.M. McCartney. (2024) (in revision) https://www.biorxiv.org/content/10.1101/2024.12.12.628281v1
A Diaphanous and Enabled-dependent asymmetric actin cable array repositions nuclei during Drosophila oogenesis
Logan, G., Chou, W-C., and B.M. McCartney. (2022) Development 149(13): dev197442. doi: 10.1242/dev.197442. Epub 2022 Jul 1. PMID: 35686626
Arc1 and the microbiota together modulate growth and metabolic traits in Drosophila
Keith, S.A., C. Bishop, S. Fallacaro, and B.M. McCartney. (2021) Development 148(15): dev195222. doi: 10.1242/dev.195222. Epub 2021 Jul 29. PMID: 34323271