Tagging BK Channels Reveals Subunit Control Functions
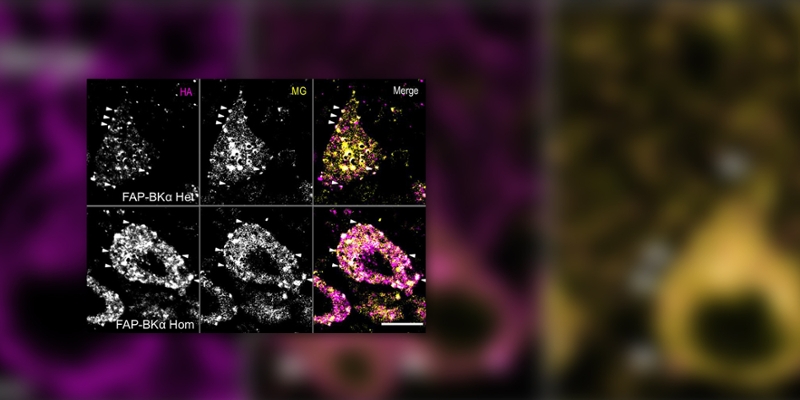
As key regulators of neuronal excitability and neurotransmitter release, large conductance voltage and calcium activated potassium channels (BK channels) are crucial in proper neuronal function. Like many proteins, BK and other ion channels are trafficked to and away from the plasma membrane, which is a key regulatory step in their function.
Using CRISPR gene targeting technology, researchers in the Bruchez and Barth labs developed a mouse model in which endogenous BK channels are tagged with fluorogen-activating peptides (FAPs), which enables fluorescent imaging of endogenous channels. Using this model, it was found that the β4 subunit of BK is involved in regulating the localization and clustering of BK channels in the Purkinje Cells of the cerebellum.
This discovery implicates this accessory subunit as crucial for regulating the localization of channels which, by their inclusion of this subunit, have distinct biophysical properties. Furthermore, the FAP system is especially well-suited for live-cell trafficking studies; thus, this model enables researchers at CMU and around the globe to study dynamic regulation of endogenous BK channel trafficking in health and disease.
Pratt CP, Kuljis DA, Homanics GE, He J, Kolodieznyi D, Dudem S, Hollywood MA, Barth AL, Bruchez MP. Tagging of Endogenous BK Channels with a Fluorogen-Activating Peptide Reveals β4-Mediated Control of Channel Clustering in Cerebellum. Front Cell Neurosci. 2017 Oct 31;11:337. doi:10.3389/fncel.2017.00337. eCollection 2017. PMID: 29163049