

Delivering the Goods
MCS Chemists Develop Polymers to Improve Drug Delivery
by Jocelyn Duffy
Imagine having a sinus infection, but instead of taking an antibiotic every few hours over the course of a few weeks, you take one pill once a week. Or that a person with cancer could take a daily pill rather than having to sit in a hospital or clinic for hours over the span of weeks to get an IV infusion of a chemotherapy drug. Or that a patient with severe burns could use a type of liquid bandage to stop the progression of the burn.
One day, these advances just might come true thanks to the clever work of polymer scientists including those at the Mellon College of Science.
Approximately 99 percent of drugs are taken orally, in the form of a pill or liquid. Unlike intravenous drugs, oral drugs need to travel through the digestive system before they can enter the blood stream and reach their target. Delivery methods must walk a delicate line between keeping the drugs from being degraded or exiting the body before they can do their work, but not allowing them to accumulate to the point where they become toxic.
“Macromolecules can take drugs that are already proven and deliver them in a better way,” said Krzysztof Matyjaszewski, the J.C. Warner University Professor of the Natural Sciences.
Many successful drugs, and promising new ones, have been developed over the past 30 years thanks to a number of advances in the biological sciences. But getting those drugs to their target cells at the right time and in the right concentrations has proven to be tricky.
“The field of biology is making so much progress, especially in making recombinant proteins and other biomolecules. We’re looking to leverage those advances in a way that’s more technology oriented,” said Newell Washburn, associate professor of chemistry and biomedical engineering.
To do that, chemists are looking to improve existing drugs’ efficacy by creating polymeric vehicles to deliver the drugs in a precise, time-sensitive manner.
In Matyjaszewski’s lab, they’re using controlled radical polymerization (CRP) methods to create precisely designed polymers, including multi-armed star polymers and multi-segmented nanogels. Using CRP, the researchers bind existing drugs to the polymers. They then create polymer chains of a specific length that are linked together with cleavable acetal or disulfide bonds. At the start, the entire complex of polymer and drug is so large that it can’t be cleared by the kidneys, but as the drug travels through the body the bonds react to changes in things like time or pH and release individual segments of the polymer piece by piece (see figure). This carefully controlled degradation allows the drug to be released into the blood stream at the optimal time and breaks the complex into smaller pieces that can be cleared through the renal system.
“Well-defined, tailor-made polymers make a big difference because they can be reproduced nicely, and we can fine-tune the properties, degradation rate and toxicity,” Matyjaszewski said.
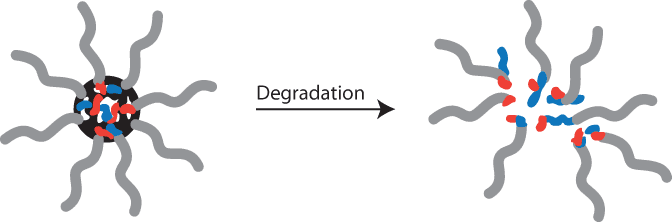
Star polymers, like the one represented here, have a core with polymeric arms radiating from the center. For drug delivery, a drug or biological molecule is at the core and the arms are connected by bonds that react to conditions like changes in pH. As the bonds react, the arms are released, allowing the drug to reach its target in a controlled manner and safely be cleared from the body.
The polymer vehicles created by Matyjaszewski are generalizable and have the potential to be used with a variety of drugs. For example, in a project funded by the Department of Defense, Matyjaszewski and Biological Sciences and Biomedical Engineering Professor Jeffrey Hollinger are using CRP techniques to create a nanostructured polymer that will precisely deliver an RNA-based therapeutic agent that can help to block painful bone ossification, a condition where bone starts to grow in the muscles and tendons at the site of an amputation.
The delivery of RNA-based therapeutics is an area of great promise for polymeric vehicles. These types of nucleic acid-based drugs, however, pose a different kind of challenge—rather than worrying primarily about how the drugs exit the body, researchers are more concerned with keeping the drugs in the patient’s system. RNA-based therapeutics need time to break through the membrane of the targeted cell, enter the cell and alter the expression of targeted genes.



From top: Kris Matyjaszewski, Subha Das and Newell Washburn
A key type of RNA therapeutics is small interfering RNA (siRNA), short strands of RNA that can be used to disrupt the expression of genes.
“Being able to block certain genes, or a mutant gene, or a viral gene is the future of medicine. But a long-standing challenge has been delivery,” said Associate Professor of Chemistry Subha Das, whose area of expertise is getting other molecules to attach to nucleic acids like DNA and RNA. “RNA chemistry is allowing us to attach molecules to RNA that weren’t possible before using straightforward methods.”
Das and Matyjaszewski have partnered to create a bioconjugate that attaches biocompatible polymers to siRNA. The polymer enables the siRNA to be more stable and less easily degraded, giving more time for the siRNA molecule to enter the cell and be effective against its cellular target.
While promising, these delivery systems still have to pass the rigors of human testing and FDA approval. It is a feat that the researchers know is hard to achieve, but the potential results are worth the risk.
“The dream of every chemist is to see molecules he or she made become useful. In biomaterials the impact potential is tremendous, but the chance of getting there is small,” Matyjaszewski said.
Of the MCS polymers being created as drug delivery systems, the closest to human testing is one being developed by Washburn to treat severe burns. But unlike Matyjaszewski and Das’s creations, Washburn’s is a topical application.
After a burn, the inflammatory system goes into overdrive in order to heal the injury. In the case of partial thickness burns—burns where the first layer of skin is totally gone, and the second layer only partially gone—the immune system overcompensates and can kill the remaining cells of the second layer of skin. This process is called burn progression and is a frequent complication of serious burns.
Most inflammatory conditions are caused by the same over-reaction of the inflammatory response, driven by the signaling molecule tumor necrosis factor alpha (TNF). TNF inhibitors have been somewhat successful in treating systemic inflammatory conditions like rheumatoid arthritis, but since they act as an immunosuppressant and degrade quickly, they haven’t been able to be used for localized inflammatory conditions. They are specifically ineffective for burns, because patients with burns need to be able to fight off infection.
Knowing that TNF inhibitors worked well against inflammation, Washburn decided to try to adapt the drug’s delivery system so it could be used to treat burns. He knew the drug would need to be applied topically so that it wouldn’t suppress the body’s immune system. But it would also need to be anchored to something so it would dissipate more slowly in order to treat burn progression. Washburn covalently bonded the TNF inhibitor to a large, biocompatible polysaccharide called hyaluronic acid.
In lab tests, he and his research team applied the hyaluronic acid/TNF bioconjugate to a burn and found that it significantly decreased inflammation and slowed burn progression. The ultimate goal would be to apply these materials to humans. If the results matched those in the lab, it would mean a quicker recovery, less scar formation and a reduced need for painful skin grafts.
Washburn is on his way to getting his bioconjugate into clinical trials. He recently formed a spinoff company called Washburn Therapeutics, which will further develop the technology to the point where it can begin to be tested in humans.
“Biomaterials give you a chance to have an impact in people’s lives,” Washburn said. “There are a lot of opportunities out there, and we’re poised to take advantage.”