Growing Fat Cells on Silk May Help Researchers Better Understand Diseases
By Marika Yang
Media Inquiries- College of Engineering
Carnegie Mellon University’s Rosalyn Abbott is analyzing fat, or adipose tissue, to learn about the characteristics of disease mechanisms and metabolic behavior. As obesity continues to be a serious health concern in the United States, more research is necessary to combat rising health risks and potential metabolic disorders.
“Our lab is focused on trying to determine what affects our metabolism in healthy and diseased states,” said Abbott, an assistant professor of biomedical engineering since August 2017. “That is the umbrella that everything else falls under, whether it's trying to create more accurate models to predict human responses, or looking at how different factors are influencing this change in metabolism during disease processes.”
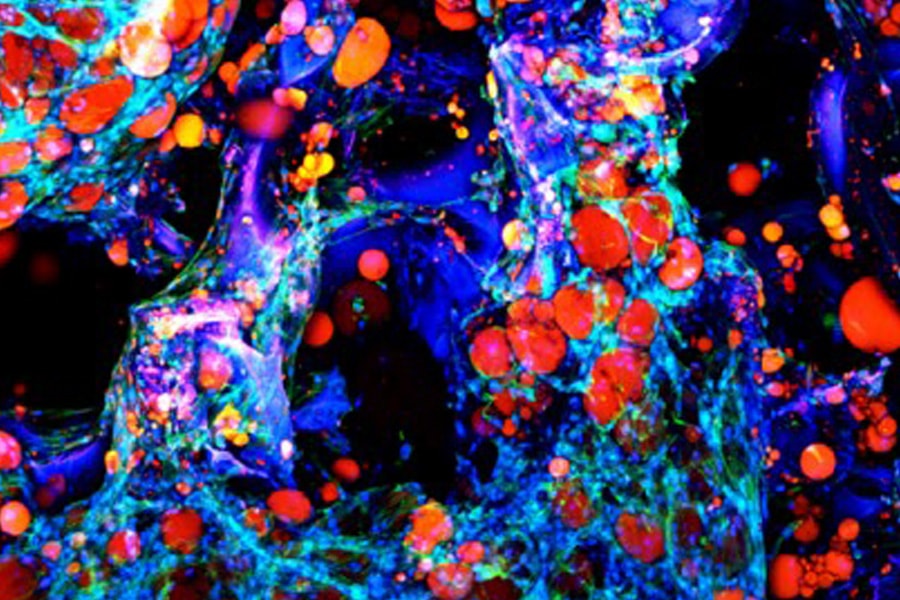
Abbott’s lab develops human adipose tissue models to explore mechanisms of different diseases. Most research about human metabolism comes from using animal tissue models, but these models don’t always translate to human disease mechanisms. Adipose tissue itself cannot maintain its structure outside the body, so Abbott and her team use silk as a scaffolding material to help sustain the tissue’s structure. Silk is a natural biomaterial that has a wide range of versatile engineering applications.
With silk scaffolds, Abbott has successfully recreated functions of adipose tissue in vitro, specifically to study the disease progression of type II diabetes. By stimulating the adipose tissue model with insulin, which type II diabetic patients are not as responsive to, Abbott can use the glucose reaction as a marker to experiment on different factors that may or may not cause changes in glucose levels.
“Our long-term goal is to use our adipose tissue model to understand what molecular factors drive the complex transition from obesity to type II diabetes,” Abbott said.
Some tests have reaped surprising results. When some stimuli are tested on the adipose tissue models using tissue samples from different patients, these stimuli lead to the same response from every sample. Other stimuli are patient-specific and produce vastly different responses.
“When I exposed tissues obtained from different patients to a drug that acted as an exercise analog, only a small percentage responded consistently with the altered metabolic response that we expected,” Abbott said.
"Our long-term goal is to use our adipose tissue model to understand what molecular factors drive the complex transition from obesity to type II diabetes.” — Rosalyn Abbott
Initially, she focused on creating a reproducible, consistent model of human adipose tissue responses. While this is still the goal of her research, the variability in responses between patients indicates that models need to be more accurate to more closely predict responses. Because individuals react to different stimuli differently, it is not possible for a single model to predict how all people respond to certain stimuli. Abbott’s research has expanded to study the backgrounds and histories of patients to predict various responses.
Another part of Abbott’s research is focused on obesogens. These chemicals don’t directly cause obesity, but instead predispose patients to accumulate excess adipose tissue — and excess weight — when those patients are exposed to them during early stages of development. An example of an obesogen is Bisphenol A, an endocrine disruptor commonly found in plastic food and drink packaging.
“I’m really interested in chemical exposures, particularly chemicals like obesogens that when exposed in utero, actually predispose you later in life, because that’s a developmental stage,” Abbott said. “It doesn’t mean necessarily that you’ll be obese, but it just means that you would be more likely to gain more weight later in life.”
Overall, her research aims to learn about how diseases develop. Because of the variability of patient responses from testing, Abbott envisions engineered adipose tissue models that are personalized and patient-specific that could be used for individual health care strategies.
“While our current goal is to gain insights into the disease mechanisms of type II diabetes, these tissue models aren’t limited to that disease processes,” Abbott said. “They could be used to inform weight loss strategies or the complex metabolic transition of other diseases that are affected by changes in the adipose tissue such as cardiovascular diseases or breast cancer.”