NEWS & Events
- September 30, 2016: Graduate student Krista Freeman is recognized at the Cleveland State University Distinguished Alumni Award Gala as the first distinguished alumna of the Jack, Joseph and Morton Mandel Honors College.
- April 29, 2016: Graduate student Ting Liu successfully defended her PhD thesis, "Biophysics of encapsidated DNA states in viruses and their role for infectivity".
- August 20, 2015: Graduate student Ting Liu selected to the inaugural class of CMU Presidential Fellows as a recipient of Bruce McWilliams Presidential Fellowship in the Mellon College of Science 2015-16.
- July 20, 2015: Carnegie Mellon University Press Release "Scientists identify achilles' heel of virus's tough outer shell."
- April 13, 2015: Graduate student Dave Bauer successfully defended his PhD thesis, "Influence of internal genome pressure on viral particle infectivity and stability".
- "Graduate Student Krista Freeman is selected to attend the 65th Lindau Nobel Laureate Meeting in Lindau, Germany." Only the 55 most qualified young scientists from the United States are given the opportunity to enrich and share the unique atmosphere of the Lindau Nobel Laureate Meetings.The meeting is held from 28 June to 3 July 2015.
- October 14, 2014: PNAS article, "Rapid transitions in viral DNA story, highlighted in PNAS In this issue front page"
- November 11, 2014: Pittsburgh Post-Gazette "Body temperature linked to DNA activity inside disease-causing virus"
- September 30, 2014: Carnegie Mellon University Press Release, "Viral Infection Might Just Be a Phase Transition"
- Graduate student Dave Bauer was awarded an NIH fellowship from MBSB graduate program based on his successful grant application.
- Listen to the popular science podcast "The Skeptics' Guide to the Universe" in the final segment, "Science or Fiction" starting around 63 min our discovery of Herpes pressure is being discussed.
- July 24, 2013: Carnegie Mellon University Press Release, "First Experimental Evidence of Internal Pressure Inside Herpes Virus".
- Graduate Student Krista Freeman was awarded a 2013 NSF Graduate Research Fellowship Program (GRFP) Fellowship.
- March 21, 2013: Graduate student Dave Bauer is awarded an Outstanding Presentation Prize at the Grad Expo 2013 at the University of Pittsburgh.
- June 2012: Professor Evilevitch has been awarded an NSF grant.
- February 2012: Graduate student Udom Sae-Ueng receives Astrid and Bruce McWilliams Fellowship for impressive research accomplishments in Physical Virology.
- July 17, 2010: Carnegie Mellon Front Page, "Physicist Makes Pivotal Discovery"
- February 17, 2010: Pittsburgh Post-Gazette article, "Researcher says physics may outsmart viruses" features our research.
- February 5, 2010: Carnegie Mellon University Press Release, "Carnegie Mellon Physicist the First To Measure Energy Released From a Virus During Infection"
- January 2010: Science Daily article, "Energy of Attacking Virus Revealed" , discusses our recent calorimetry findings.
January 2010 - "High-powered living DNA cannon" Nano Tsunami - Nano Medicine in depth, 2005
- "DNA Goes Ballistic" Science,
July 2003 - "Some viruses faster than a speeding bullet" ABC News,
July 2003
Herpes Virology Research
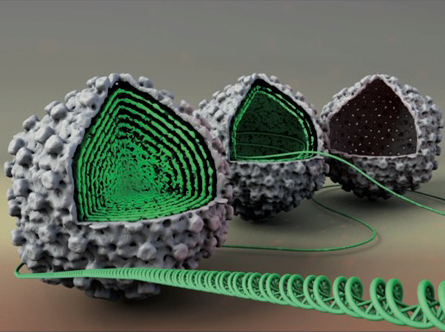
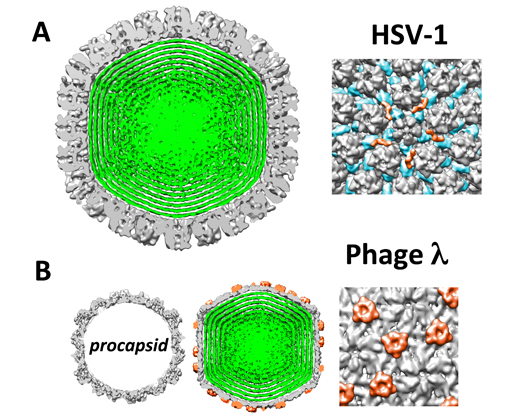
Herpesviruses are a leading cause of human viral disease, second only to influenza and cold viruses. Herpesviruses consist of a double-stranded (ds) DNA genome contained within a protein shell, termed the capsid, that is surrounded by an unstructured protein layer (the tegument) and a lipid-envelope. During viral replication, an ATP-dependent motor packages the genome into a preformed capsid through a unique opening created by the portal complex. Herpes Simplex virus type 1 (HSV-1) is a prototypical model system to study the general infection mechanisms of herpesviruses and other viruses that release their genome into the cell nucleus without capsid disassembly. We have recently shown that HSV-1 genome packaging creates an internal pressure of tens of atmospheres within the viral capsid. This pressure results from bending stress and repulsive forces acting on the tightly packaged DNA molecule. We also found that despite its liquid crystalline state inside the capsid, the DNA is fluid-like which facilitates its ejection into the cell nucleus during infection. The fluidity or, equivalently, mobility of the closely packaged DNA strands caused by interstrand repulsive interactions is regulated by the ionic environment of the cellular cytoplasm.
Between rounds of replication, the virion must be sufficiently stable to ensure that the packaged genome is retained within the capsid. Conversely, during infection the virion must be unstable enough to allow genome release into the cell nucleus. A precise balance between these physical aspects of the viral capsid and its encapsidated genome is crucial to the viral replication cycle. Using HSV-1 as our primary model system, we investigate the roles of intracapsid DNA mobility, internal DNA pressure and capsid stability for viral replication with respect to retention of the packaged genome inside the capsid and its subsequent ejection during infection. These studies provide new insights into the key mechanisms facilitating as well as inhibiting viral infectivity.
Physical Virology of Herpesviruses and Phage
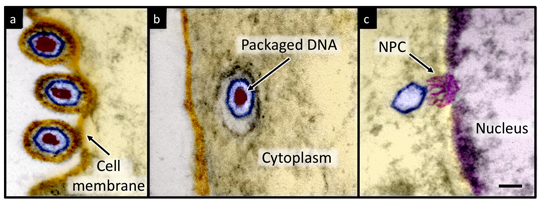
 Viruses are simple lifeless entities that cannot reproduce on their own and therefore depend on host cells to provide them with the necessary life support mechanisms. Simplified, all viruses consist of a protein shell (capsid) that protects the viral genome (DNA or RNA). To infect, the viral genome must enter the cell, where it hijacks the host cell’s machinery and synthesizes multiple copies of virions. This can lead to cell lysis, which is a lethal event.
Viruses are simple lifeless entities that cannot reproduce on their own and therefore depend on host cells to provide them with the necessary life support mechanisms. Simplified, all viruses consist of a protein shell (capsid) that protects the viral genome (DNA or RNA). To infect, the viral genome must enter the cell, where it hijacks the host cell’s machinery and synthesizes multiple copies of virions. This can lead to cell lysis, which is a lethal event.
Physical virology is a rather new field that seeks to define the physical mechanisms controlling virus development. This knowledge can provide information essential to the rational design of new anti viral strategies with less specificity for a limited number of viruses. Furthermore, biological and physical simplicity relative to other biological systems have made viruses an attractive physical model system to study fundamental prosperities of DNA compaction and translocation as well as protein self-assembly using viral capsids.
Our Biophysical Tools
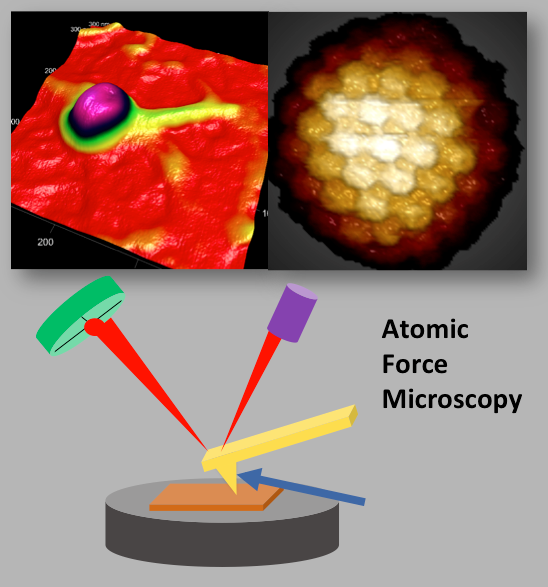
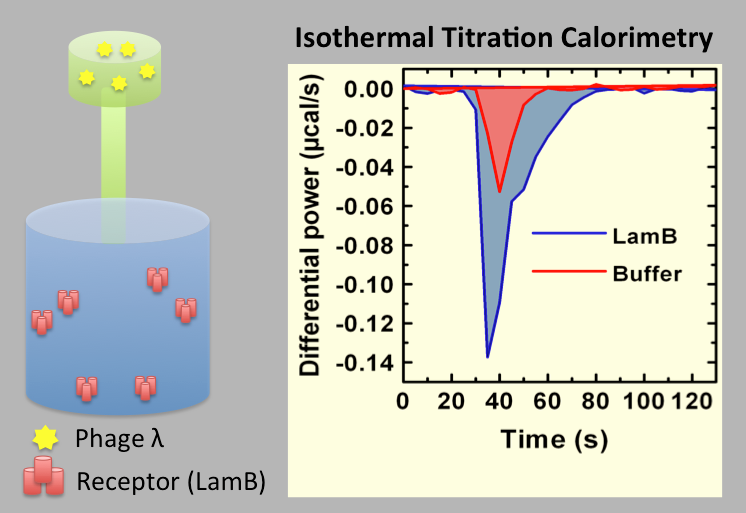
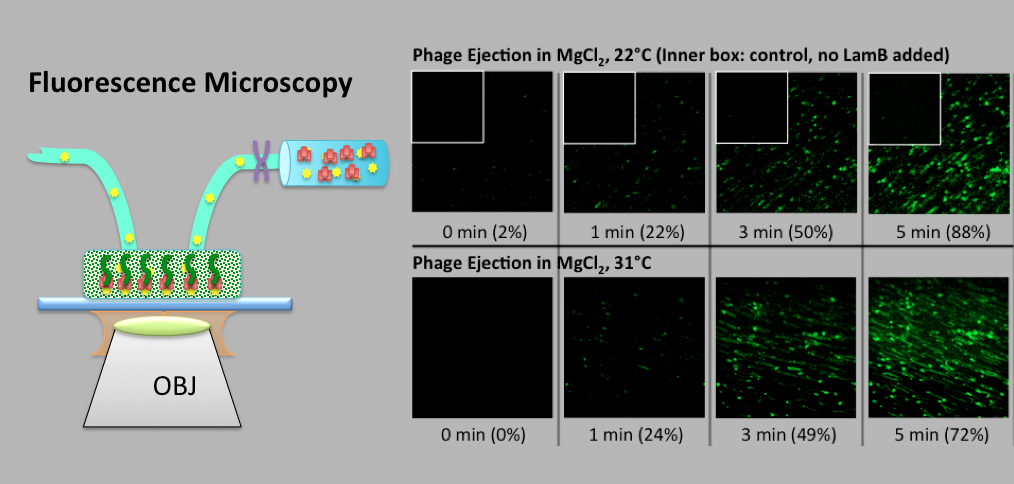
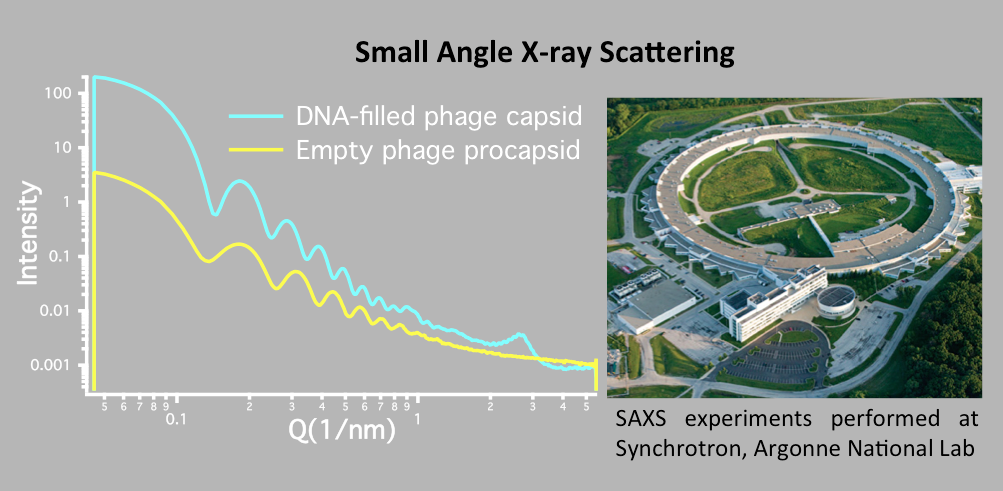
Evilevitch's physical virology group investigates fundamental physical principles that control viral encapsidation and genome release. Our group has discovered a way to determine genome pressure in viral capsid and found that to be as high as 20 atm in human Herpes Simplex Type 1 virus, a pressure equivalent to that at a depth of 600 ft in the ocean. We are specifically interested in determining the physical nature of genome packaging and release, the kinds of pressures involved, the strengths and elastic properties of the capsids, and limits on the amount of material that can be encapsidated. The main tools in the lab are: Atomic Force Microscopy (AFM), microcalorimetry, high resolution cryo electron microscopy and single molecule fluorescence, small angle x-ray scattering and light scattering.