Enhanced Steam-Coal Gasification Kinetics in a CaO/KOH Chemical Looping Gasifier: Experimental Results Published
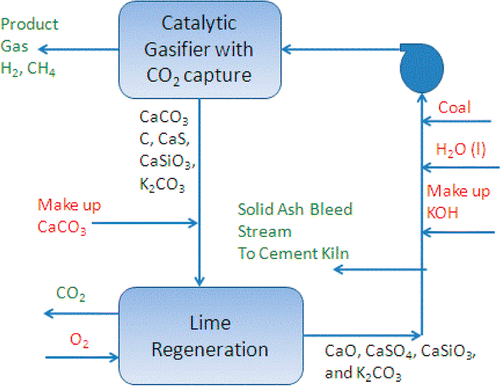 A process flow diagram of one of the cycles studied in the Energy & Fuels paper.
A process flow diagram of one of the cycles studied in the Energy & Fuels paper.
Energy & Fuels has published research from the TPES group regarding steam-coal gasification with in-situ carbon capture. This research is a collaboration between TPES and the National Energy Technology Laboratory (DOE/NETL).
TPES researcher Nicholas Siefert conducted experimental studies of coal gasification using calcium oxide (CaO) as the main dual-functioning catalyst-capture agent. It was demonstrated that adding small amount of potassium hydroxide (KOH) can increase the steam-coal gasification kinetics 4-fold compared to only using CaO and 10-fold compared with no CaO or KOH.
Using two different coal types and temperatures between 700ºC and 900ºC, Siefert studied the effect of these catalyst-capture agents on (1) the syngas composition, (2) CO2 and H2S capture, and (3) the steam-coal gasification kinetic rate. The dry syngas composition from the gasifier was roughly 20% methane, 70% hydrogen, and 10% other species when a CaO:C molar ratio of 0.5 was added. The CaO and KOH species also capture H2S and HCl that may be in the feedstock, making this a potentially viable process for converting high sulfur coals or chlorine containing wastes, such as MSW, into syngas to generate electricity with near-zero emissions of acid and greenhouse gases.
In addition to the gasification experiments, Siefert et al. conducted multi-cycle studies in which the CaCO3 was calcined by heating to 900ºC to regenerate the CaO, which was then re-used in repeated CaO-CaCO3 cycles. The increased steam-coal gasification kinetics rates for both CaO and CaO+KOH persisted even when the material was reused in six cycles of gasification and calcination.
This research was also presented at the 2012 AIChE Conference in Pittsburgh, PA.
This work was published in the journal Energy & Fuels in a special issue: Accelerating Fossil Energy Technology Development through Integrated Computation and Experimentation. It is available online with DOI 10.1021/ef302192p, or in our publications section.